 
The data used to obtain CE certification of these devices, however, are not in the public domain. All tested devices are on the market in Europe based upon studies conducted by each of the manufacturers. Of the seven devices that failed all have the CE marking. This is a concern as the CE marking is a manufacturer’s certification documenting the accuracy, reliability and safety of devices. ConclusionsĪ high percentage of devices failed accuracy testing in this study. The devices that passed the tests were the Smart Peak Flow® and the Mini Wright®. Of the nine devices that were tested, two passed and seven failed. Three CE guideline tests were utilised, and standards require that all three tests are passed for the device to obtain certification. 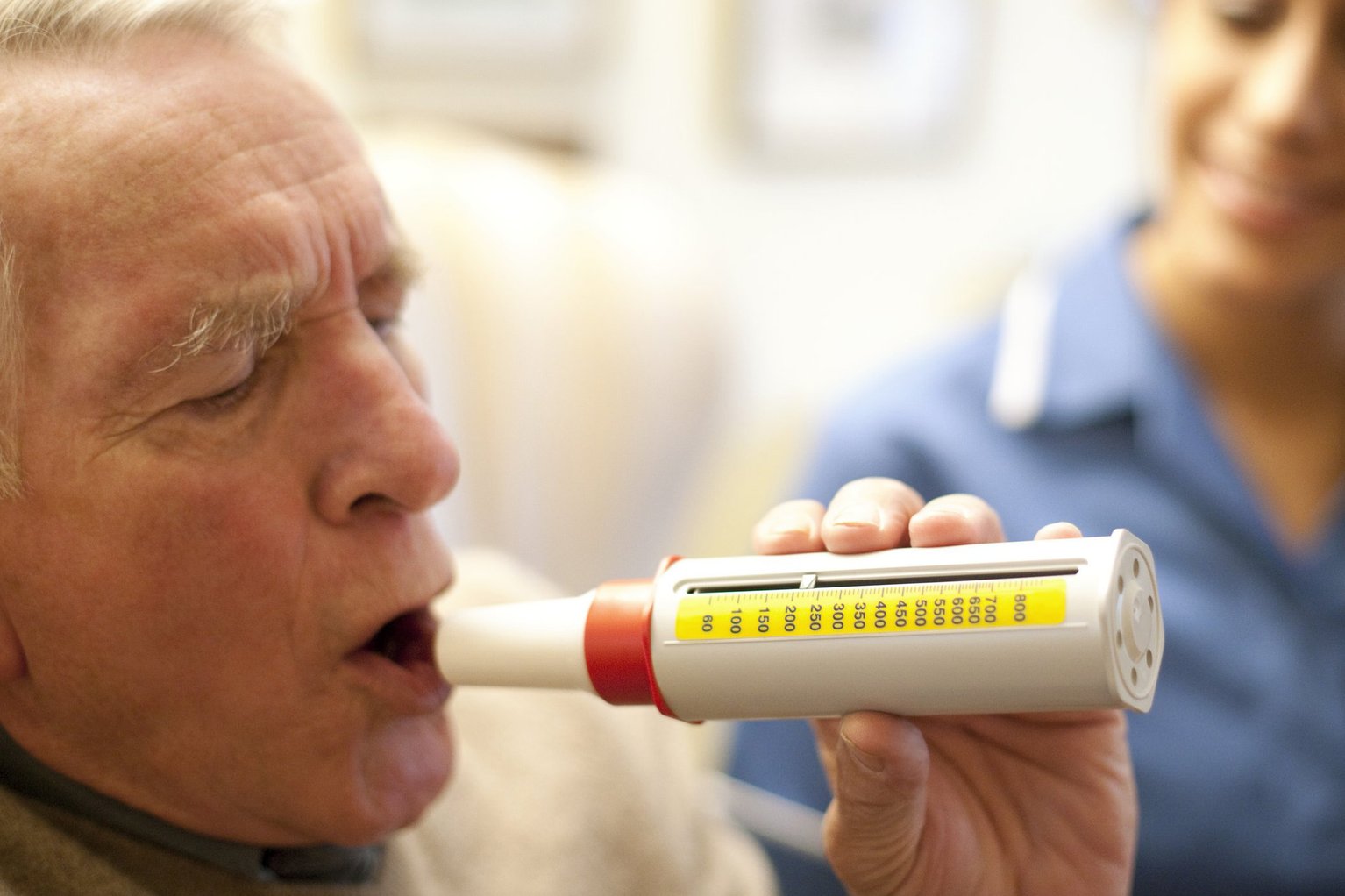
Using predefined flow (time) and volume (time) waveforms, peak flow meters and spirometers were tested for validation and calibration. 
The apparatus chosen was the pulmonary waveform generator manufactured by Piston Medical Ltd. This study was performed with a certified piston pump testing apparatus. All standards, including accuracy, from these guidelines must be met to obtain CE marking. The devices were bench tested following the ISO 23747:2015 accuracy guidelines for medical devices measuring peak flow. The nine devices were selected as they all had received or were in the process of receiving CE approval in Europe and were readily obtainable. The CE marking is a manufacturer’s declaration that their product complies with European health regulations and it is a requirement for marketing medical devices in Europe. The study evaluates and compares the accuracy of nine peak flow meters (“PFMs”) and spirometers that are currently available in Europe and have Conformité Européene (“CE”) marking. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
